 Overall, Architector represents a transformative step towards cross-periodic table computational design of metal complex chemistry.ĭata-driven methods for materials 1, 2, 3 and chemical 4, 5 discovery have enjoyed considerable success, fueled by the availability of accurate high-throughput structure-based simulation approaches 6, 7, 8, 9. Further, we demonstrate out-of-the box conformer generation and energetic rankings of non-minimum energy conformers produced from Architector, which are critical for exploring potential energy surfaces and training force fields. 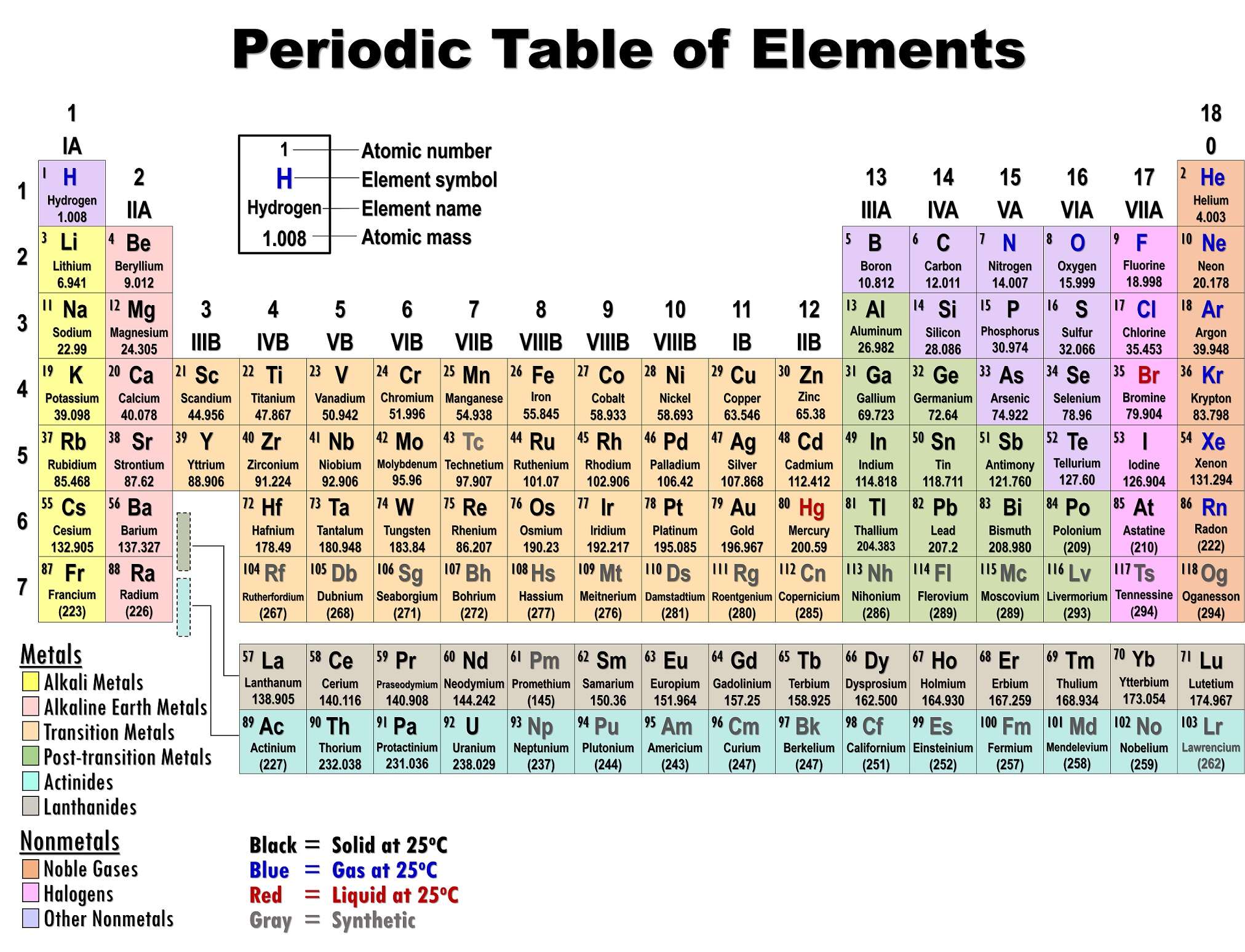
Over a set of more than 6,000 x-ray diffraction (XRD)-determined complexes spanning the periodic table, we demonstrate quantitative agreement between Architector-predicted and experimentally observed structures. Architector leverages metal-center symmetry, interatomic force fields, and tight binding methods to build many possible 3D conformers from minimal 2D inputs including metal oxidation and spin state. Beyond known chemical space, Architector performs in-silico design of new complexes including any chemically accessible metal-ligand combinations. Here, we introduce Architector, a high-throughput in-silico synthesis code for s-, p-, d-, and f-block mononuclear organometallic complexes capable of capturing nearly the full diversity of the known experimental chemical space. 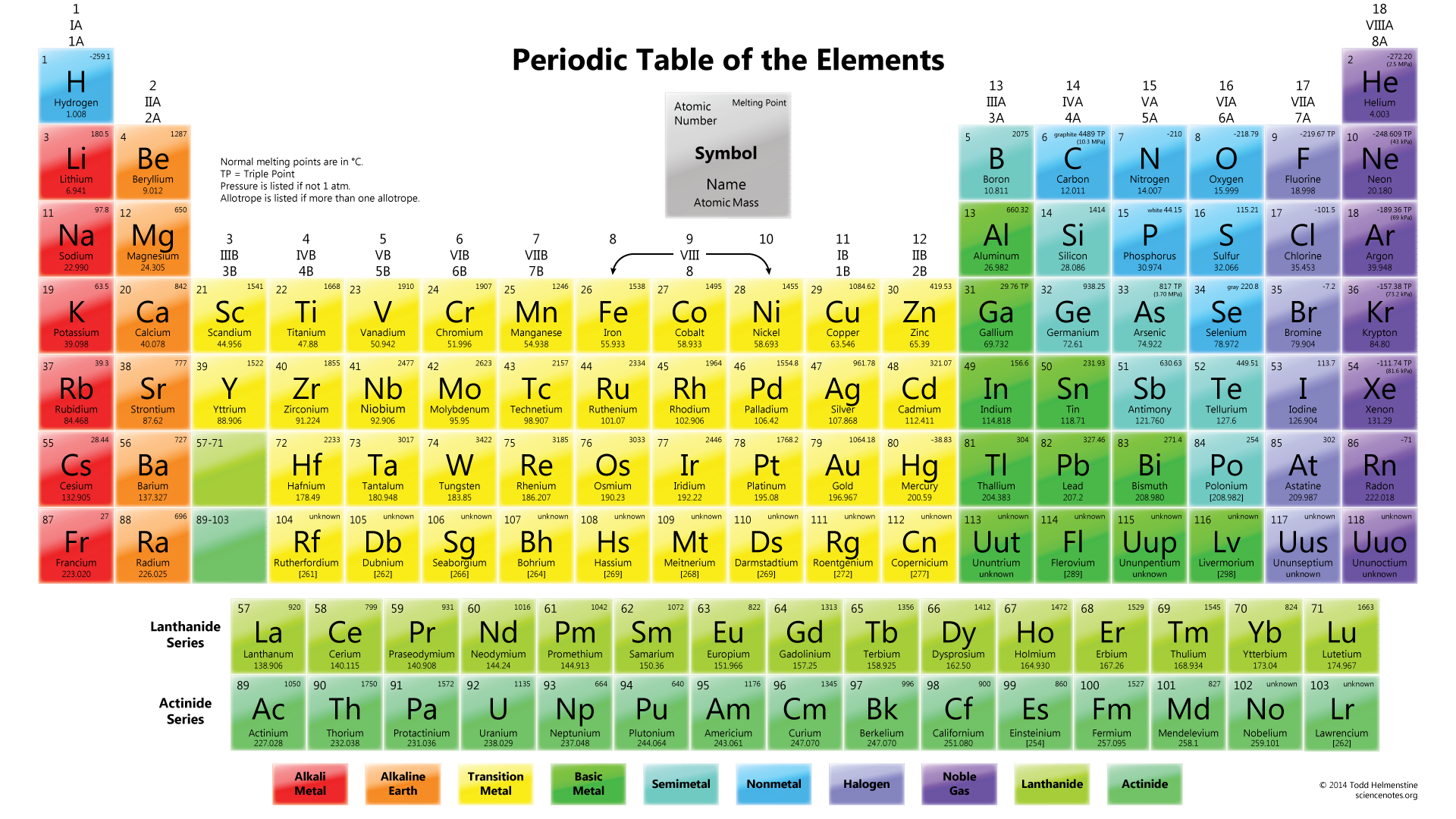
Three-dimensional (3D) structural generation and prediction for these organometallic systems remains a challenge, limiting opportunities for computational chemical discovery. The periodic table as a list of elements arranged so as to demonstrate trends in their physical and chemical properties.Rare-earth and actinide complexes are critical for a wealth of clean-energy applications.Describe and model the structure of the atom in terms of the nucleus, protons, neutrons and electrons comparing mass and charge of protons neutrond and electrons. Use the Periodic Table to predict the ratio of atoms in compounds of two elements. Unit 1: Structures, Trends, Chemical Reactions, Quantitative Chemistry and Analysis.Unit C1: Structures, Trends, Chemical Reactions, Quantitative Chemistry and Analysis.(g) elements being arranged in order of increasing atomic number and in groups and periods in the modern Periodic Table, with elements having similar properties appearing in the same groups.2.2 ATOMIC STRUCTURE AND THE PERIODIC TABLE.(h) elements being arranged in order of increasing atomic number and in groups and periods in the modern Periodic Table, with elements having similar properties appearing in the same groups.1.2 ATOMIC STRUCTURE AND THE PERIODIC TABLE.Unit 1: CHEMICAL SUBSTANCES, REACTIONS and ESSENTIAL RESOURCES.(a) elements being arranged according to atomic number in the Periodic Table.Unit 1: THE LANGUAGE OF CHEMISTRY, STRUCTURE OF MATTER AND SIMPLE REACTIONS.The Periodic Table can be used to determine whether an element is a metal or non-metal.Atomic structure and bonding related to properties of materials.Elements are arranged in the periodic table in order of increasing atomic number. 
RSC Yusuf Hamied Inspirational Science Programme.Introductory maths for higher education.The physics of restoration and conservation. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
